Materials
BCA protein assay (Pierce Biotech. cat. #23225)
Small-PROTEAN II gel apparatus (Biorad)
Costar gel-loading tips (Krackler Scientific, cat. #MN520R-LRS)
Prestained SDS-Page broadrange molecular weight standard (NEB, cat. #P7708S)
Trans-blot Semi-Dry Transfer cell (Biorad)
Immobilon-P membrane (Millipore, cat. #IPV00010)
heat-sealable bags (Kapak, cat. #TRS-95250)
Kodak Biomax light autoradiagraphy film, 13 x 18 cm (Perkin Elmer Existence Sciences, cat. #868-9358)
Radtape (Diversified Biotech, cat. #RAD-10)
Reagents
TEMED (Sigma)
Tween-20 (Sigma)
Cost Chopper non-fat dry milk
Isopropanol (Sigma)
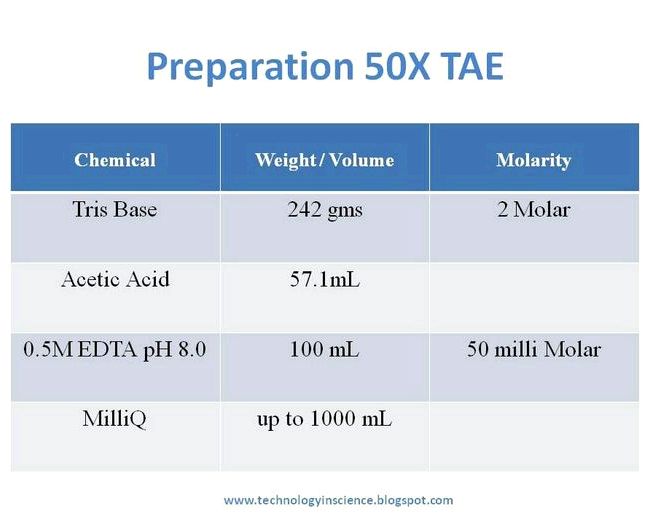
Gel Buffers
Acrylamide solution (30% acrylamide/.8% bis-acrylamide)
-dissolve 30g acrylamide (FW=71.08), .8g bis-acrylamide (FW=154.17) inside a total amount of 100ml water. Filter through .2um filter.
4X Running Gel Buffer (1.5M Tris, pH 8.8)
-dissolve 36.3g Tris Base (FW=121.1) in 150ml water. Adapt to pH 8.8. Add water to total amount of 200ml. Filter through .2um filter.
4X Stacking Gel Buffer (.5M Tris, pH 6.8)
-dissolve 12.1g Tris Base (FW=121.1) in 150ml water. Adapt to pH 6.8. Add water to total amount of 200ml. Filter through .2um filter.
10% SDS
-dissolve 10g SDS (FW=288.38) inside a total amount of 100ml water.
10% Ammonium Persulfate
-dissolve 1.0g APS (FW=228.2) inside a total amount of 10ml water. Store at -20C in 50-100ul aliquots.
1M Tris, pH 6.8
-dissolve 12.1g Tris Base (FW=121.1) in 80ml water. Adapt to pH 6.8. Add water to total amount of 100ml. Filter through .2um filter.
6X SDS Sample Buffer (.375M Tris pH 6.8, 12% SDS, 60% glycerol, .6M DTT, .06% bromophenol blue)
-combine 3.75ml 1M Tris-Cl, pH 6.8, 6ml glycerol, 1.2g SDS (FW=288.38), .93g DTT (FW=154.2), 6mg bromophenol blue. Add water to total amount of 10ml. Store at -20C in .5ml aliquots.
2X SDS Sample Buffer (.125M Tris pH 6.8, 4% SDS, 20% glycerol, .2M DTT, .02% bromophenol blue)
-combine 2.5ml 4X Tris pH 6.8 (stacking gel buffer), 2ml glycerol, 4ml 10% SDS (FW=288.38), .31g DTT (FW=154.2), 2mg bromophenol blue. Add water to total amount of 10ml. Store at -20C in .5ml aliquots.
1X SDS Sample Buffer (.0625M Tris pH 6.8, 2% SDS, 10% glycerol, .1M DTT, .01% bromophenol blue)
5X Electrophoresis Buffer (.125M Tris, .96M glycine, .5% SDS)
-dissolve 30.3g Tris Base (FW=121.1), 144.1g glycine (FW=75.07), and 10g SDS (FW=288.38) inside a total amount of 2L water.
1X Electrophoresis Buffer (.025M Tris, .192M glycine, .1% SDS)
-add 200ml 5X electrophoresis buffer to 800ml water.
Western Blot Buffers
Transfer Buffer (.048M Tris, .039M glycine, 20% methanol, .00375% SDS)
-dissolve 11.64g Tris Base (FW=121.1), 5.86g glycine (FW=75.07) in
1500ml water. Add .750ml 10% SDS (FW=288.38). Add 400ml methanol. Add water to total amount of 2L. No pH adjustment necessary.
10X TBST Wash Buffer (.1M Tris HCl, 1.5M NaCl, .5% Tween-20)
-dissolve 31.52g Tris HCl (FW=157.6), 175.32g NaCl (FW=58.44) in
1900ml water. Add 10ml Tween-20. Adapt to pH 7.2. Add water to total amount of 2L.
1X TBST Wash Buffer (.01M Tris HCl, .15M NaCl, .05% Tween-20)
-add 100ml 10X TBST to 900ml water.
5% milk Blocking Buffer
-Dissolve 5g milk inside a total amount of 100ml 1X TBST.
Method
- Determine cell phone number or protein concentration to become loaded onto gels and just what volume to load per well. Note max volumes for various comb configurations.
- Block membrane in 20ml blocking buffer overnight at 4°C or 1h at 70 degrees on rocker inside a seal-a-meal bag.
- Dilute Ab in blocking buffer (1:1000 for serum or 5ug / 10ml for purified Ab). Incubate blots with Ab for 1h at rom temperature or overnight at 4°C on rocker inside a seal-a-meal bag.
- Wash blots with 50ml 1X TBST on orbital shaker 3 occasions, a few minutes each.
- Dilute HRP conjugated Ab 1:10,000. Incubate blots with secondary Ab for 1h at 70 degrees or overnight at 4°C on rocker inside a seal-a-meal bag.
- Wash blots with 50ml 1X TBST on orbital shaker 3 occasions, a few minutes each.
- Add 5ml chemiluminescent substrate to every blot, incubate for five minutes. Place blots among transparency film. Push out air bubbles. Develop Western blot. Perform a one minute contact with check signal intensity. Continue longer or shorter exposures when needed.
(Max. exposure length = 60 min. Min exposure length = 1 sec.) - For top backgrounds, wash in TBST then re-develop.